The mass of any substance per unit volume of a material is known as absolute density (d). 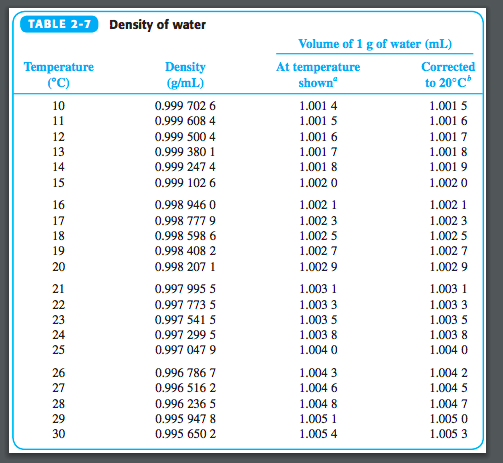
A high density object is one that is hefty and compact. Density can also be defined as the quantity of mass per unit of volume. You can try some simple experiments and observations like mixing oil and water, put ice cubes in warm water, and so on.Density:- Density is a term that describes how much space (volume) an object or substance takes up in relation to the amount of stuff it contains (its mass). Now that you know and understand what water density is, we highly suggest you conduct some experiments at home so that you can apply and better grasp the things you just learned. What is its density? Is it gold? Should you buy it? You can view the answer here Conclusion You measure the cube and find that it is 2 cm on each side, and weighs 40 g. You pull out your old geology text and look up gold in the mineral table, and read that its density is 19.3 g/cm3. The person wants you to buy it for $100, saying that is a gold nugget. Problem 7: A golden-colored cube is handed to you. If a rock needs to weigh 2,000 kilograms (about 2 tons) in order not to be shifted by waves, how big (what volume) does it need to be? You are using basalt, which has a typical density of 3200 kg/m3 Problem 6: Rocks are sometimes used along coasts to prevent erosion. It is made of granite, which has a typical density of 2.8 g/cm3. It is 30 centimeters on each side, and so has a volume of 27,000 cm3. Problem 5: You decide you want to carry a boulder home from the beach. Problem 4: In the above two examples which rock is more dense? which is less dense? Problem 3: In the above two examples which rock is heavier? Which is lighter? Problem 2: You have a different rock with a volume of 30cm3 and a mass of 60g. 
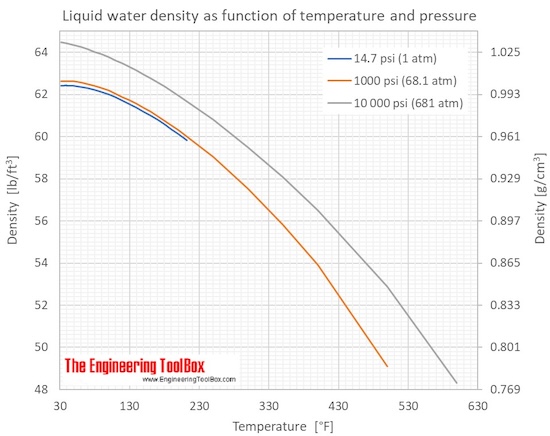
Problem 1: You have a rock with a volume of 15cm3 and a mass of 45 g. Using the equation and the information we discussed above, try to answer some of these practice problems from Carleton College’s website. The data below is from Harper College’s website. You would then have to make sure that your sample is always equilibrated to the calibrated temperature of the pycnometer. With that, measurement is only valid at that certain temperature. Keep in mind that most pycnometers are calibrated for use in certain temperatures. The density is calculated based on this formula Density = (M2 − M1)/Flask Volume. It affects the density of the water because mass also increases every time you put salt in it.īy simply referring to the formula we cited before, we can already infer that salinity does affect water salinity.Ī pycnometer is usually made up of glass and a ground stopper. So when the water froze and turns into ice, it becomes denser as compared to when it was in its liquid state. In the picture above, ice floats in water.Īs we glossed over a few times, water temperature alters the water density. The water density table below is from metrologia Temp./ CĮven though ice is the solid-state of water, it is less dense than liquid water. If you look at the table below, you would see that the weight and density of the water slightly change as temperature changes. So when you boil water, it would slightly get lighter. Oil is less dense than water (below 1) so it is no wonder why it floats on water.Īs we have pointed out before, the temperature can alter the density of water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
